Last updated on May 20, 2026

Controlled Substance Inventory Log free printable template
pdfFiller is not affiliated with any government organization
Why pdfFiller is the best tool for your documents and forms
End-to-end document management
From editing and signing to collaboration and tracking, pdfFiller has everything you need to get your documents done quickly and efficiently.
Accessible from anywhere
pdfFiller is fully cloud-based. This means you can edit, sign, and share documents from anywhere using your computer, smartphone, or tablet.
Secure and compliant
pdfFiller lets you securely manage documents following global laws like ESIGN, CCPA, and GDPR. It's also HIPAA and SOC 2 compliant.

What is Controlled Substance Inventory Log
The Controlled Substance Inventory Log is a healthcare form used by medical facilities to track the inventory of controlled substances.
pdfFiller scores top ratings on review platforms




I only used one form, the Statement of Net Worth form. It was simple and easy to use.
PDF Filler is everything I hoped it would be. Made this job very easy.
Who needs Controlled Substance Inventory Log?
Explore how professionals across industries use pdfFiller.
Comprehensive Guide to Controlled Substance Inventory Log
What is the Controlled Substance Inventory Log?
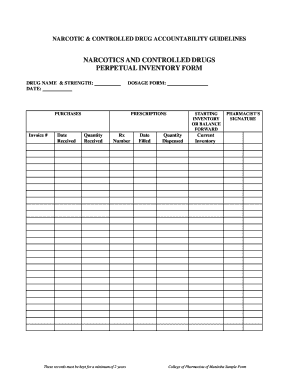
The Controlled Substance Inventory Log is a critical document used in healthcare settings to ensure accurate tracking of controlled substances. This log plays an essential role in maintaining compliance with regulations and enhancing accountability among healthcare providers. Key details recorded in the log include the substance name, quantity, and other relevant information necessary for proper inventory tracking.
Maintaining a controlled substance inventory log is vital for preventing misuse and ensuring proper handling of these substances. By keeping accurate records, healthcare facilities can uphold safety standards and respond effectively during audits or inspections.
Purpose and Benefits of the Controlled Substance Inventory Log
The primary benefit of using a controlled substance inventory log is to facilitate compliance with healthcare regulations. Accurate inventory tracking not only ensures safety but also reinforces accountability among personnel handling these substances. This practice is essential for preventing misuse and mitigating the risk of diversion.
Healthcare organizations that utilize this inventory log can expect improved operational effectiveness and enhanced safety protocols. The log serves as a useful tool during inspections, as it provides a clear record of how controlled substances are managed.
Who Needs the Controlled Substance Inventory Log?
Various healthcare professionals and organizations are required to maintain a controlled substance inventory log. Pharmacies and hospitals are primary users, as they regularly handle these substances. Additionally, regulatory agencies closely monitor controlled substance usage, making it crucial for these entities to keep accurate logs.
Situations such as audits and inspections necessitate the use of this inventory log. By having accessible records, healthcare providers can quickly demonstrate compliance with regulations.
How to Fill Out the Controlled Substance Inventory Log
Completing the controlled substance inventory log involves several key steps to ensure accuracy and compliance. First, provide the substance name, followed by its strength and form. Next, record the date the substance was received, including the name of the distributor and the invoice number. Finally, document the amount received along with the initials of the staff member responsible for the entry.
To avoid common pitfalls, adhere to best practices such as double-checking entries for completeness and accuracy. Individuals should also ensure the log is updated regularly and stored securely.
Submission Methods for the Controlled Substance Inventory Log
Upon completing the controlled substance inventory log, users have several submission options. These include digital submission via email or other electronic platforms, as well as physical delivery methods. It is crucial to choose a method that ensures the confidentiality and security of sensitive information.
Understanding the specific submission requirements is important, as some healthcare facilities may have established protocols to follow when submitting these logs.
Security and Compliance for the Controlled Substance Inventory Log
When managing the controlled substance inventory log, security and compliance play a pivotal role. Healthcare organizations must implement robust security measures to protect sensitive information. Features such as access controls and encryption are essential for safeguarding these records.
Moreover, compliance with regulations like HIPAA and GDPR is imperative when handling medical records. Protecting sensitive information ensures that healthcare facilities can maintain a trustworthy environment for patients and staff alike.
Using pdfFiller for Your Controlled Substance Inventory Log
pdfFiller is a powerful tool that can streamline the process of managing the controlled substance inventory log. With its intuitive features, users can easily fill out, edit, and eSign the form from any web browser. The platform offers enhanced capabilities for sharing and managing this critical document.
Utilizing pdfFiller not only simplifies the log completion process but also ensures that sensitive information is handled securely and efficiently.
Final Steps After Completing the Controlled Substance Inventory Log
After filling out the controlled substance inventory log, it is essential to review the document thoroughly for accuracy before submission. Tracking the submission and confirming its receipt is also advisable. Additionally, retaining a copy of the log is crucial for compliance purposes, as it serves as a record for future audits or inquiries.
By following these final steps meticulously, healthcare providers can ensure they maintain the highest standards in inventory management.
How to fill out the Controlled Substance Inventory Log
-
1.To access the Controlled Substance Inventory Log on pdfFiller, visit the pdfFiller website and use the search function to find the form by its name.
-
2.Once you locate the form, click on it to open it in the pdfFiller interface, which allows you to view and edit the document easily.
-
3.Before filling out the form, gather all necessary information, including the names of controlled substances, details regarding their forms, strengths, sizes, dates received, distributor names, and invoice numbers.
-
4.Begin completing the form by clicking into each blank field and entering the corresponding information. Use the toolbar for any additional tools you may need to fill out the form seamlessly.
-
5.Be sure to check each entry for accuracy, ensuring that the details are correct and clearly written to prevent any misunderstandings.
-
6.Once all fields are filled, review the entire log for any mistakes or missing information, making adjustments as needed before finalizing the document.
-
7.To save your completed form, click the Save button. You can also download it as a PDF or submit it electronically through your preferred method using pdfFiller’s submit options.
Who should use the Controlled Substance Inventory Log?
The Controlled Substance Inventory Log is designed for healthcare providers, pharmacies, and clinical staff involved in the management and documentation of controlled substances.
What information do I need to complete the log?
To effectively complete the Controlled Substance Inventory Log, you’ll need details about each controlled substance, including its name, form, strength, size, and distributor information.
What are the submission methods for this log?
You can submit the Controlled Substance Inventory Log electronically through pdfFiller or print it for manual submission to your facility’s regulatory body or for record-keeping purposes.
Are there deadlines for submitting the log?
Deadlines for submitting the Controlled Substance Inventory Log can vary depending on your healthcare facility's policies or regulatory requirements, so it's essential to check with your administrator.
What are common mistakes to avoid when filling out this log?
Common mistakes include failing to accurately record all details, forgetting to initial entries, and not verifying the information before submission. Always double-check your entries.
How often should the inventory log be updated?
The Controlled Substance Inventory Log should be updated regularly, particularly after receiving new stock, dispensing medications, or as required by your facility’s inventory policies.
What should I do if I make a mistake on the form?
If you make a mistake on the Controlled Substance Inventory Log, simply erase the incorrect information using the editing tools on pdfFiller, and make the necessary corrections before finalizing the form.
Related Catalogs
If you believe that this page should be taken down, please follow our DMCA take down process
here
.
This form may include fields for payment information. Data entered in these fields is not covered by PCI DSS compliance.